Viral Vector CDMO Services & Viral Platform Manufacturing Support
Viral vectors operate as biological delivery systems.
They do more than carry genetic material. They must enter the right cells, protect or deliver the payload, support the intended expression or activity, remain stable through manufacturing and storage, and meet safety, impurity, and potency requirements. A vector program therefore needs manufacturing built around delivery function, not only particle production.
This modality group includes AAV, lentiviral vectors, adenoviral vectors, retroviral vectors, HSV vectors, oncolytic viruses, baculovirus systems, viral vaccine vectors, helper-virus systems, replication-defective vectors, replication-competent viral platforms, engineered capsids, dual-vector systems, hybrid viral vectors, and specialized viral delivery platforms.
Each platform changes the CDMO route.
AAV does not route like lentivirus. Lentivirus does not route like adenovirus. An oncolytic virus does not route like a replication-defective vector. A baculovirus expression system does not use the same control logic as a clinical gene therapy vector. A viral vaccine vector carries different development and supply requirements from an ex vivo cell therapy vector input.
CDMO Network supports viral vector and viral platform programs across plasmid input planning, producer cell systems, transient transfection, stable producer lines, infection-based production, upstream vector production, harvest, clarification, nuclease treatment, downstream purification, concentration, buffer exchange, sterile processing, analytical development, potency assays, formulation, frozen storage, fill-finish coordination, regulatory CMC, GMP readiness, tech transfer, scale-up, and clinical or commercial supply planning.
Our support includes early feasibility material, research-grade vectors, preclinical supply, GMP clinical vector manufacturing, commercial readiness, vector inputs for cell therapy, in vivo gene therapy vectors, viral vaccine vectors, oncolytic platforms, and specialty viral systems.
Viral vector manufacturing succeeds when the product retains delivery function, safety profile, purity, stability, and usable supply characteristics from production through final use.
Viral vector history and why the field requires specialized CDMO support
Viral vectors became important because viruses naturally evolved to deliver genetic material into cells. Scientists adapted that biology for research, gene therapy, vaccines, oncology, cell therapy, and engineered medicine. Early gene transfer work helped define how retroviral and adenoviral vectors could move genetic material into cells. Later advances in AAV, lentiviral systems, helper systems, producer cell technologies, and purification methods expanded the field into modern therapeutic development.
The field also learned quickly that vector manufacturing is not standard biologics manufacturing with a different product name.
Vectors combine biology, payload design, producer systems, process timing, particle formation, impurity control, infectious or functional performance, and storage sensitivity. The process can produce measurable particles while still delivering weak biological function. The batch can show strong genome titre and still underperform in potency testing. A purification method can improve purity while reducing infectivity. A freeze-thaw cycle can preserve appearance while damaging function.
That history explains why viral vector CDMO strategy needs platform-specific execution.
The manufacturing route must account for the viral system, payload, intended use, analytical package, safety testing, potency logic, formulation, and downstream clinical or commercial pathway.
AAV vector manufacturing
AAV programs represent one of the most technically demanding areas of viral vector manufacturing.
AAV routes can involve transient transfection, producer cell systems, helper-virus systems, adherent or suspension production, HEK293 platforms, Sf9 and baculovirus-based systems, capsid engineering, tissue tropism design, self-complementary AAV, dual AAV systems, and novel serotypes.
CDMO Network supports AAV programs across plasmid input control, producer system selection, upstream production, harvest, clarification, nuclease treatment, affinity or chromatography-based purification, empty/full analysis, concentration, buffer exchange, formulation, frozen storage, fill-finish coordination, and GMP manufacturing.
AAV quality control requires more than total vector genomes.
Our support includes genome titre, capsid titre, empty/full ratio, potency assays, transduction assays, infectivity or functional readouts where relevant, residual host-cell DNA, residual plasmid DNA, host-cell protein, residual nuclease, aggregation, purity, identity, sterility coordination, endotoxin, bioburden, and stability testing.
AAV development also needs capsid-specific thinking. Different serotypes and engineered capsids can behave differently in production, purification, aggregation, potency, and stability. A route that works for one capsid does not automatically transfer to another.
AAV manufacturing works best when capsid, payload, production system, analytics, and formulation are developed as one connected route.
Lentiviral vector manufacturing
Lentiviral vectors play a major role in cell therapy, ex vivo gene modification, and some gene therapy workflows.
Lentivirus manufacturing can involve plasmid inputs, producer cells, transient transfection, stable producer systems, upstream culture, harvest timing, clarification, concentration, purification, sterile filtration where appropriate, frozen storage, and final formulation.
Lentiviral vectors are sensitive products.
Envelope stability, infectious titre, transduction efficiency, vector copy number, potency, residual impurities, shear exposure, filtration conditions, freeze-thaw behavior, and storage conditions all affect performance. A lentiviral vector can lose functional value even when physical indicators appear acceptable.
CDMO Network supports lentiviral programs across research-grade vectors, preclinical vectors, GMP clinical supply, cell therapy vector inputs, stable producer development, process development, purification, analytics, formulation, cryostorage, and supply planning.
Our support includes infectious titre, functional titre, p24 or capsid-related assays where relevant, genome copy testing, transduction efficiency, vector copy number support, residual plasmid testing, host-cell DNA, host-cell protein, sterility coordination, endotoxin, mycoplasma, replication-competent lentivirus testing coordination, and stability studies.
Lentiviral vector routes must protect transduction performance.
The useful vector is the vector that modifies cells reliably under the intended process conditions.
Adenoviral, retroviral, HSV, and specialized viral platforms
Viral vector programs extend well beyond AAV and lentivirus.
Adenoviral vectors, retroviral vectors, HSV vectors, poxviral vectors, VSV-related systems, measles vectors, alphavirus platforms, helper-virus systems, and hybrid viral vectors all create different manufacturing and quality requirements.
Adenoviral programs often require control over infectivity, particle-to-infectious ratio, residual host-cell DNA, potency, and storage. Retroviral vectors require producer system control, infectious titre, safety testing, and use-case-specific analytics. HSV vectors require large-genome handling, potency, safety-related testing, and platform-specific purification logic. Viral vaccine vectors require antigen expression, potency, identity, impurity control, and scalable supply planning.
CDMO Network supports specialized viral platforms by routing each program to partners with relevant platform experience, containment fit, analytical capabilities, and GMP or non-GMP quality systems aligned to the product’s intended use.
Our support includes upstream production, infection-based processes, producer cells, purification, concentration, buffer exchange, formulation, safety-related testing coordination, potency assay development, and regulatory CMC support.
Specialized viral platforms need specific manufacturing logic.
The route must match the viral biology and the product use.
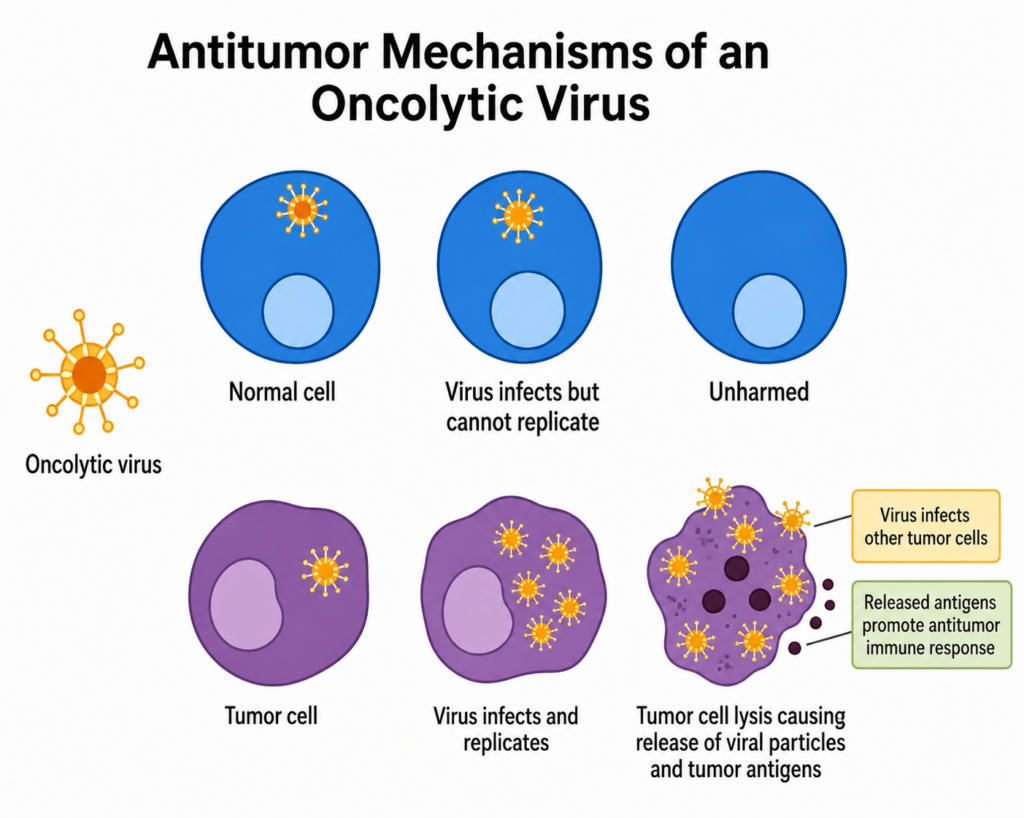
Oncolytic viruses and replication-competent platforms
Oncolytic viruses and replication-competent viral systems require strong control over viral behavior.
These programs can include replicating oncolytic viruses, conditionally replicating vectors, engineered HSV systems, adenovirus-based oncolytic platforms, vaccinia-related systems, VSV-related systems, and other tumor-targeting viral products.
Manufacturing must protect identity, infectivity, replication behavior, potency, purity, safety profile, and formulation stability. These programs can require specialized containment, platform-specific cell substrates, infection control, harvest timing, purification, residual impurity testing, and robust functional assays.
CDMO Network supports oncolytic and replication-competent viral programs through platform routing, process development, GMP readiness, potency strategy, infectivity testing, safety-related testing coordination, formulation, fill-finish coordination, and regulatory CMC planning.
These products often need assays that reflect mechanism. A particle count alone does not explain replication behavior or tumor-cell activity. Potency, infectivity, identity, and functional performance must connect to the intended biological mechanism.
Replication-competent platforms require more than capacity.
They require facilities, systems, and quality controls aligned to viral behavior.
Baculovirus systems and viral production platforms
Baculovirus systems support both production and product use.
They can support recombinant protein expression, VLP production, vaccine antigen manufacturing, AAV production in insect-cell systems, and baculovirus-based research or industrial applications.
A baculovirus route can involve Sf9 cells, High Five cells, baculovirus stock generation, infection optimization, harvest timing, recombinant protein production, VLP assembly, vector-related production, purification, and stability testing.
CDMO Network supports baculovirus and insect-cell platform programs across research-scale expression, process development, GMP-oriented production, protein and antigen manufacturing, VLP-related workflows, and AAV production routes where appropriate.
Our support includes infection process development, multiplicity of infection optimization, cell density strategy, harvest timing, clarification, purification, characterization, potency or activity testing, formulation, and scale-up.
Baculovirus systems are powerful because they support complex biological production.
They still require control over infection dynamics, product quality, impurity profile, and process timing.
Plasmid inputs and producer cell systems
Viral vector manufacturing often depends on critical starting materials.
Plasmids, helper constructs, producer cells, packaging systems, master cell banks, working cell banks, viral seeds, and helper-virus systems can determine vector productivity, impurity profile, consistency, and timeline.
CDMO Network supports vector programs by coordinating plasmid DNA manufacturing, producer cell strategy, banking, input quality, supplier qualification, sequence confirmation, endotoxin control, residual impurity awareness, and documentation alignment.
Plasmid problems can become vector problems. Weak topology, late delivery, high endotoxin, residual RNA, incomplete sequence documentation, or inconsistent quality can affect transfection performance, vector yield, residual impurity burden, and batch repeatability.
Producer cell systems also matter. Cell history, passage control, growth behavior, transfection efficiency, infection dynamics, bank characterization, and culture performance can all affect final vector quality.
Vector manufacturing begins before the production run.
It begins with the inputs.
CDMO Network drives analytical development and vector characterization
CDMO Network delivers comprehensive analytics that accurately measure physical quantity, biological function, purity, safety, and stability of viral vectors.
We provide:
- Genome, particle, capsid, infectious, functional, and transduction titres
- Potency and expression assays
- Empty/full ratio analysis
- Vector copy number determination
- Identity testing, sequencing, and impurity profiling (residual host-cell DNA, plasmid DNA, proteins, nuclease, helper virus, replication-competent virus, endotoxin, bioburden, mycoplasma)
- Aggregation analysis and stability-indicating methods
AAV programs rely on precise capsid data and full/empty ratios. Lentiviral programs demand high infectious titres and transduction efficiency. Oncolytic and vaccine vectors require robust infectivity, replication control, antigen expression, and safety testing.
Formulation, fill-finish, and frozen handling
CDMO Network designs formulation strategies that actively protect vector potency and function against temperature, shear, freeze-thaw, aggregation, and container interactions.
We offer expert support in cryoprotectant screening, buffer optimization, surfactant evaluation, container compatibility, in-use and shipping studies, and post-thaw performance.
Our fill-finish services include sterile vial filling, controlled freezing, visual inspection, labeling, packaging, and cold-chain coordination — all designed to maintain functional titre and clinical usability.
GMP manufacturing, tech transfer, and scale-up
CDMO Network executes reliable scale-up while safeguarding vector identity, purity, and potency.
We support research-grade, preclinical, GMP clinical, and commercial manufacturing for in vivo gene therapy, cell therapy vectors, viral vaccines, and oncolytic viruses.
Our end-to-end capabilities include process development, upstream and downstream optimization, analytical development, release testing, quality documentation, tech transfer, comparability studies, and full lifecycle support.
CDMO Network ensures successful scale-up by addressing shifts in transfection efficiency, impurity profiles, empty/full ratios, and recovery — delivering consistent, high-quality vectors from bench to commercial supply.
Viral vector niches supported
This modality group includes mainstream and highly specialized viral platforms.
Support extends across AAV, lentiviral vectors, adenoviral vectors, retroviral vectors, HSV vectors, oncolytic viruses, baculovirus systems, viral vaccine vectors, helper-virus systems, replication-defective vectors, replication-competent viral platforms, AAV gene therapy vectors, novel capsid AAV, capsid-engineered AAV, dual AAV systems, lentiviral gene therapy vectors, integration-deficient lentiviral vectors, HSV gene therapy systems, replicating oncolytic viruses, conditionally replicating viral vectors, adenovirus gene therapy platforms, hybrid viral vectors, baculovirus expression systems, viral production platforms, and research-to-GMP vector transitions.
Each program receives a route based on viral platform, payload, production system, intended use, safety requirements, analytical burden, formulation need, scale, and supply model.
A single vector manufacturing template cannot cover the full field.
The viral biology and use case define the route.
바이럴 벡터 제조는 단순한 입자 생산이 아니라 전달 기능, 순도, 안전성, 안정성, 역가를 함께 관리하는 작업입니다. AAV, 렌티바이러스, 아데노바이러스, HSV, 온콜리틱 바이러스, 바큘로바이러스 시스템은 각각 다른 생산 방식과 분석 전략을 요구합니다. CDMO Network는 플라스미드 입력물, 생산 세포, 업스트림 공정, 정제, 역가 시험, 잔류 불순물 관리, 제형, 냉동 보관, GMP 제조, 기술이전과 스케일업을 벡터의 실제 사용 목적에 맞춰 연결합니다.
Requirements for high-quality viral vector CDMO services
CDMO Network builds robust viral programs by first aligning platform, payload, producer system, intended use, quality attributes, potency, safety, formulation, scale, and supply strategy. We support a wide range of viral platforms through complete upstream production, downstream purification, potency and infectivity testing, impurity removal, formulation development, stability studies, GMP manufacturing, fill-finish, tech transfer, scale-up, and clinical-to-commercial supply. Programs demand precise capsid control with full/empty analysis, high infectious titres with strong transduction efficiency, rigorous identity and safety testing, plus effective antigen expression and manufacturing scalability. We deliver vectors with intact identity, powerful delivery function, high purity, proven safety, reliable potency, and long-term stability from initial production through final supply.
Learn more about here about –> CDMO Network Cell Line Development Services
Contact our team at info@cdmonetwork.com
